From Compliance Burden to Competitive Edge: How Digital Transformation Is Reshaping the Pharmaceutical Industry
The pharmaceutical industry has always operated under pressure. But the pressure today feels different — regulatory requirements are tightening, global supply chains are more fragile than they appear, and the race to bring new therapies to market faster has never been more intense. All at the same time.
For manufacturers and healthcare operators across Asia-Pacific, the question is no longer whether to digitally transform. It is whether the current operational setup can keep absorbing the cost of not doing so.
The Challenges Facing Pharma Today
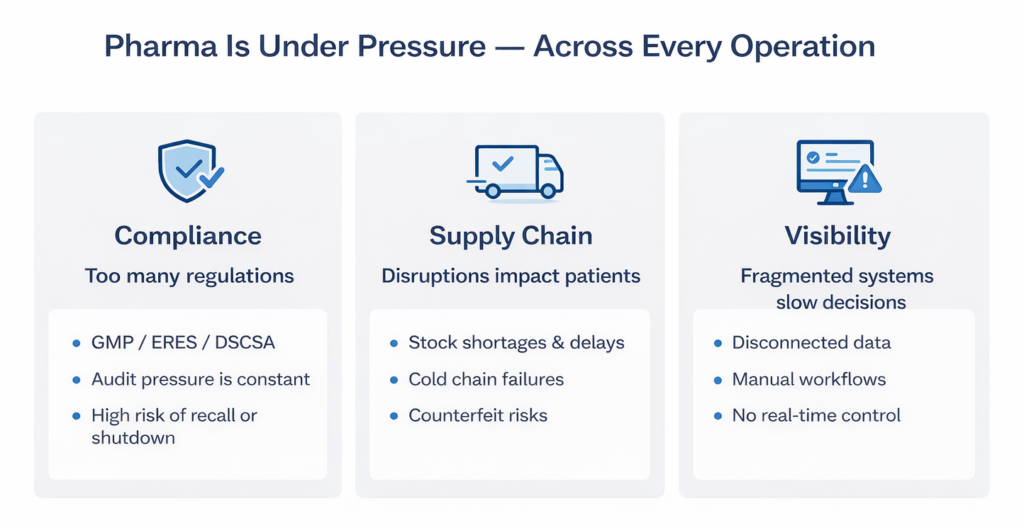
1. Regulatory Compliance Is Getting More Complex — Not Less
ERES) regulations, GMP standards, serialization mandates such as the EU Falsified Medicines Directive (FMD) and the US Drug Supply Chain Security Act (DSCSA) — these are no longer distant concerns for manufacturers operating in or exporting to regulated markets. They are daily operational realities.
Pharmaceutical compliance violations have cost the industry over $50 billion since 2000. And the risk is not just financial: a gap in the audit trail, an unresolved CAPA, or a batch traceability failure can halt production, trigger a recall, or result in regulatory action.
For companies still relying on paper-based workflows and disconnected systems, audit readiness is a significant operational cost — not a standard outcome.
2. Supply Chain Disruptions Have a Direct Impact on Patients
As the COVID-19 pandemic made clear, pharmaceutical supply chain failures are not just an operational problem — they determine whether the right medicine reaches the right patient on time and at the right quality. Counterfeit drugs, stock shortages, and treatment delays are the real-world consequences of supply chains that lack end-to-end visibility.
The pharmaceutical supply chain spans raw material sourcing, manufacturing, packaging, cold chain logistics, and last-mile distribution. Each stage carries distinct compliance obligations and quality risks. End-to-end visibility, powered by real-time data, enables proactive identification of issues — supplier delays, quality deviations, cold chain excursions — before they escalate. In the case of recalls, it allows rapid identification and removal of affected batches.
Without that visibility, disruptions are discovered reactively — after the impact has already reached operations or patients.
3. AI and Data Are Redefining What’s Possible
Leading pharmaceutical companies are accelerating investment in data, digital, and AI across commercial and operational functions. The use cases are no longer pilots — they are running in live production environments:
Demand forecasting: AI analyzes historical data, market trends, and seasonal patterns to reduce stockouts and overstock — both critical in pharmaceutical operations.
Predictive maintenance: Sensor data from manufacturing equipment is analyzed to anticipate failures before they cause production downtime.
Batch release acceleration: Automated quality checks and digital workflows reduce cycle times and compliance effort — STEMCELL Technologies reported an 86% reduction in time for SAP system validation for GxP guidelines after implementing RISE with SAP (Source: SAP).
Supply chain risk management: AI-driven scenario planning helps companies navigate disruptions before they escalate into production or distribution failures.
The ERP Foundation: Why It Matters More Than Ever
The technologies above — AI, IoT, serialization, real-time analytics — can only deliver value when they run on a solid operational foundation. For most pharmaceutical manufacturers, that foundation is the ERP system.
A modern, cloud-based ERP purpose-built for life sciences integrates batch management, quality management, supply chain, procurement, and financial controlling on a single platform. This means:
- Every production run is traceable from raw material to batch release
- GxP documentation is embedded in workflows, not added after the fact
- Compliance reporting is automated, not manually assembled before audits
- Finance and operations work from the same data, in real time
Without this integration, digital initiatives remain isolated — IoT data in one system, quality records in another, financial reporting in a spreadsheet. The result is a patchwork that fails precisely when it matters most.
What This Means for Manufacturers in Asia-Pacific
The pharmaceutical sector in Asia-Pacific is accelerating in both directions simultaneously. Markets across the region are growing rapidly — driven by rising healthcare demand, aging populations, and increasing consumer spending on health. At the same time, regulatory expectations are rising: GMP standards, serialization requirements, and data integrity obligations are aligning more closely with EU and US benchmarks.
For manufacturers with significant dependence on imported raw materials and APIs, supply chain resilience is not a theoretical concern — it is a strategic priority. Companies that invest now in building a connected operational backbone — from shop floor to regulatory submission — will be positioned to compete on quality, speed, and cost as the market continues to differentiate between those who lead on technology and those who follow.
How TDI APJ Supports Pharmaceutical and Healthcare Companies
At TDI APJ, we implement SAP solutions for pharmaceutical manufacturers and healthcare operators across the Asia-Pacific region — with a consulting team trained and certified in pharmaceutical industry standards.
That means we do not just configure software to technical specifications. We understand how a batch release decision is made, what a CAPA workflow looks like in practice, and where compliance gaps typically surface in a pharmaceutical production environment.
Our implementations cover:
- Batch management and end-to-end serialization
- GMP compliance and quality management
- Manufacturing and production planning
- Clinical and commercial supply chain
- Procurement and supplier quality control
- Warehouse and cold chain management
- Finance, cost control, and audit trail
- Healthcare operations and scheduling
Real-world results show how digital transformation delivers measurable impact in pharmaceutical operations — from faster validation to more efficient system testing.
Key Takeaways
Regulatory compliance and supply chain continuity are the two defining operational pressures for pharmaceutical companies right now
AI and integrated data platforms are moving from pilot to production — delivering measurable results in batch release speed, forecasting accuracy, and proactive compliance
A modern, cloud-based ERP is the foundational layer that makes these capabilities possible — not an add-on
For manufacturers in Asia-Pacific, the current investment cycle is a pivotal window to modernize before regulatory requirements tighten further
Want to Go Deeper?
We have compiled a full report on digital transformation in the pharmaceutical and life sciences industry — covering regulatory trends, SAP solution capabilities, and real-world implementation outcomes across Asia-Pacific.
Leave your details below to receive the full report directly in your inbox.
Sources: SAP RISE with SAP for Life Sciences; SAP Life Sciences Industry Report; Pharma Supply Chain 2.0 — Supply Chain Wizard; ZS Pharmaceutical Trends; Pharmaceutical Technology — Keys to Navigating the Complex Drug Supply Chain; HITLAB — Digital Transformation in Pharma Supply Chain



